Ease of Use
Meeting Your Patient Needs
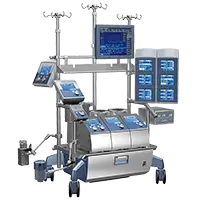
The LivaNova 3T™ Heater-Cooler System provides 3 circuits for fast and efficient heating and cooling to meet your patient and cardioplegia temperature needs.1
Compressor-based cooling and 3 independent water tanks eliminate the need for ice and water supply in the operating room, providing separate settings for warm cardioplegia, cold cardioplegia and patient temperature.1
Patient temperature settings can be adjusted to the tenth of a degree for accurate temperature control. To facilitate cleanup, a water evacuation feature allows pulling back water from the lines into the tanks at the end of a procedure.1
1 Data on file.
Resources
LivaNova Heater-Cooler System 3T Quick Reference Guide
3T Heater-Cooler Operating Instructions (October 2025)
3T Aerosol Collection Set Instructions for Use
Correction Letter, Availability of New 3T Heater-Cooler System Operating Instructions and Reminder about Design Upgrade, February 24, 2020
Vacuum and Sealing Upgrades Customer Letter, July 25, 2019
Hydrogen Peroxide Monitoring and Design Upgrade Customer Letter, October 2018
Deep-Cleaning Service Customer Letter, April 2018
US 3T Customer Letter, December 2016
US 3T Field Safety Notice Update, October 13, 2016
US 3T FAQs, October 13, 2016
US CDC Morbidity and Mortality Weekly Report, October 13, 2016
US CDC Health Advisory No. 397, October 13, 2016
US FDA Safety Communication, October 13, 2016
US 3T Certificate of Medical Necessity Form, October 2016
US 3T Customer Communication Update, July 2016
US 3T FAQs, EuroSurveillance Article, June 2016
US 3T Customer Letter, January 2016
US 3T Field Safety Notice Update, August 2015
US 3T Field Safety Notice, June 2015
Safety Information
Important Safety Information for 3T Heater Cooler
1. INDICATIONS FOR USE / INTENDED PURPOSE
EU: In accordance with the applicable regulations, the heater-cooler is used with a Stöckert/Sorin Heart-Lung Machine (S3/SC/S5/C5) for the controlled heating/cooling of 3 water circuits during extracorporeal perfusion. The water circuits are used for cooling/heating blood (in the oxygenator), hypothermia blankets or cardioplegic solutions.
US: The Heater-Cooler System 3T is used to circulate water through heat exchangers to warmor cool a patient during cardiopulmonary bypass procedures lasting 6 hours or less.
2. CONTRAINDICATIONS
There are no known contraindications for the Heater-Cooler System 3T. The attending physician is solely responsible for the use of the system.
3. WARNINGS
It is important to follow the cleaning and disinfection process described in the operating instructions to help reduce the risk of contamination. Personnel and preparation:
- Only individuals trained to use the heater-cooler should perform maintenance on the heater-cooler.
- Only service technicians authorized by LivaNovamay repair the heater-cooler system.
- Only refrigerant systems experts are authorized to repair the cooling circuit.
- Do not service the internal components of the heater-cooler. Only LivaNovarepresentatives —or other service personnel deemed qualified by LivaNova—are authorized to maintain, service, and repair internal components of the heater-cooler.
Materials and protective equipment:
- When performing any maintenance activities on the heater-cooler, follow your hospital’s personal protection equipment (PPE) policy to protect against exposure to hazardous materials, such as bacteria and/or disinfectant chemicals.
Do not connect the heater-cooler to a heart-lung machine’s auxiliary outlet. The heater-cooler should be connected to a separate dedicated AC line. To avoid the risk of electric shock, the heater-cooler must only be connected to a supply mains with protective earth.
Operation of the heater-cooler without an applied vacuum source will stop the collection of aerosol from the water tanks.
Do not position the heater-cooler’s and portable vacuum source’s (if applicable) exhaust flow toward the operating field. Position the exhaust flow away from the operating field and toward the exhaust vent system.
Operating the heater-cooler with the vacuum level indicated on the tank vacuum gauge in the red solid zone limits the aerosol collection set’s ability to collect aerosol from the water tanks.
Do not empty the external circuits until the patient has been discharged from the OR environment to reduce the potential of exposure to aerosol.
Do not shut off the applied vacuum source until you have drained the circuits and powered off the heater-cooler. Operate the heater-cooler with an applied vacuum source to ensure that aerosol continues to be collected from the water tanks.
Do not close all valves at once when draining water circuits. Always close one circuit valve at a time. Failure to close the external water circuit valves in the order specified below may result in increased levels of aerosol emission.
The aerosol collection canister contents are potentially hazardous due to collection of bacteria. Use appropriate Personal Protection Equipment (PPE), and handle the contents accordingly.
Do not use disinfectants in the water circuits during a surgical intervention. Only use disinfectants preoperatively and postoperatively.
Use only one disinfectant and make sure it is the same one you added when filling the tanks for disinfection. The use of both products will potentially result in a dangerous chemical reaction.
Do not use the heater cooler if the control panel is flashing. This means the heater-cooler is in service mode. Power off the heater-cooler and power it on again to exit service mode.
4. PRECAUTIONS/CAUTIONS
Federal law (U.S.A.) restricts this device to sale by or on the order of a physician. For a complete listing of precautions/cautions please refer to the Instructions for Use which accompany each product
The devices should be used by qualified and skilled personnel, able to follow the indications and instructions for use contained in the information provided by the manufacturer. Please contact us through our website (www.sorinmanuals.com) to receive instructions for use containing full prescribing information including indications, contraindications, warnings, precautions and adverse events.
The information contained in this summary represents partial excerpts taken from the product labeling. The information is not intended to serve as a substitute for a complete and through understanding of the device nor does this information represent full disclosure of all pertinent information concerning the use of this product. Not approved in all geograhies. Consult your labeling.
The HC3T Heater-Cooler System uses refrigerant R-513A (filling charge: 0.93 kg; Global Warming Potential: 573), in accordance with Regulation (EU) 2024/573.
Legal Manufacturer:
LivaNova Deutschland
Lindberghstrasse 25
D-80939 Munich, Germany
Distributed in the USA by:
LivaNova USA
14401 W 65th Way
Arvada, CO 80004

Essenz™ Perfusion System
Enter the New Era of Perfusion
Built on LivaNova’s 50-year legacy of safety and reliability, Essenz supports the Perfusionist in doing what is best for each patient, and allows the entire heart team to continuously improve their clinical practice.
Please Note: Essenz information available only in selected geographies.