*Curated by Orrum Clinical Analytics based on clinical literature and industry guidelines. The CoreInsights for Essenz product and LivaNova do not provide medical advice.
CoreInsights for Essenz
A new era of perfusion analytics
LivaNova and Orrum Clinical Analytics have partnered to drive forward a New Era of Analytics with CoreInsights for Essenz. This innovative portal harmonizes technology, registries, and analytics to drive continuous improvement.

Equipment Meets Data to Revolutionize Patient Care
With technology advancements and an increased focus on patient outcomes, perfusion and the role of the perfusionist are changing and entering a New Era. With automated data submission and actionable insights, CoreInsights for Essenz supports evidence-based decision-making, helping clinicians elevate the standard of care.
AUTOMATIC
Enabling AUTOMATIC, continuous data transfer for improved accuracy and workflow
CURATED
Providing CURATED, clinically relevant analytics* to measure adherence to hospital and national benchmarks
ACTIONABLE
Empowering perfusionists with ACTIONABLE, timely insights on practice patterns and key metrics
PATIENT-TAILORED
Supporting management of advanced, PATIENT-TAILORED quality initiatives with Essenz Patient Monitor

What You Can Do with CoreInsights for Essenz
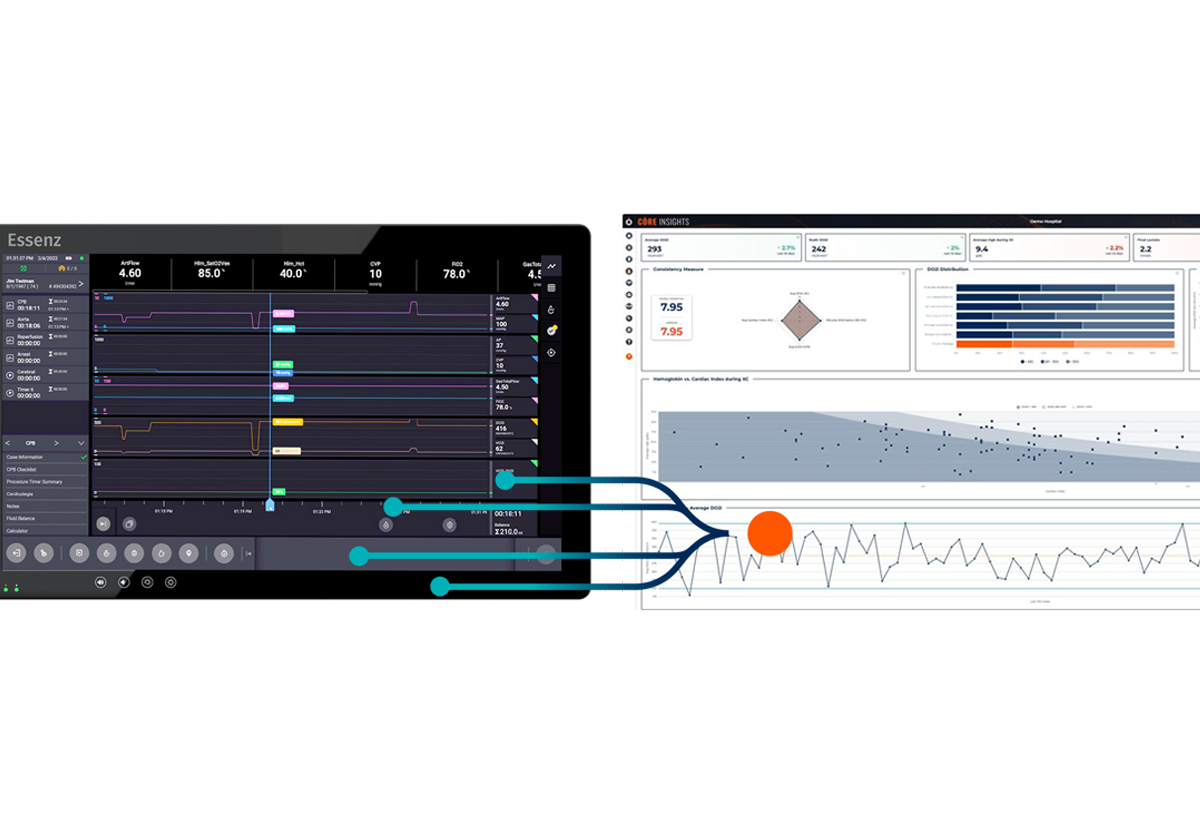
Streamline analytics execution with automatic data capture and transfer
CoreInsights for Essenz is designed to safely and automatically transfer perfusion case data from the Essenz Patient Monitor to a portal, where seamless analysis and benchmarking is conducted.
Enabled by automated case data transfer, CoreInsights for Essenz provides actionable insights and benchmarking to support evidence-based decision-making, helping clinicians elevate perfusion practice.
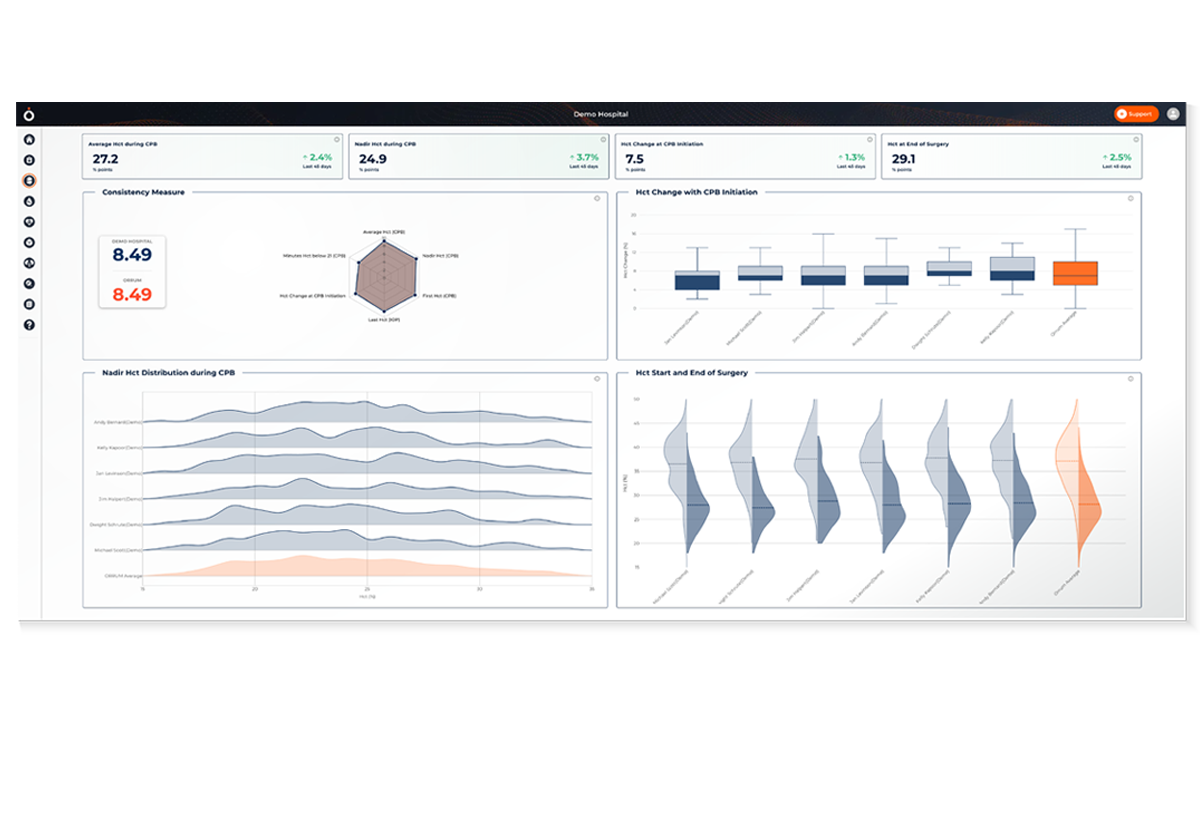
Gain insights on practice and key performance indicators through curated dashboards and reports
View summaries of all patient encounters, identifying your team’s successes and improvement opportunities based on these essential domains:
- Hemodilution
- Oxygen delivery
- Arterial pressure management
- Glycemic control
Identify special cause variation (outliers) and analyze relevant clinical information such as:
- Preoperative risk factors
- Procedural information
- Laboratory information
- Intra-operative fluid management
- Anticoagulation therapy
- CPB conduct and patient hemodynamics
Explore deeper analysis to understand associations between intraoperative practices and postoperative outcomes.
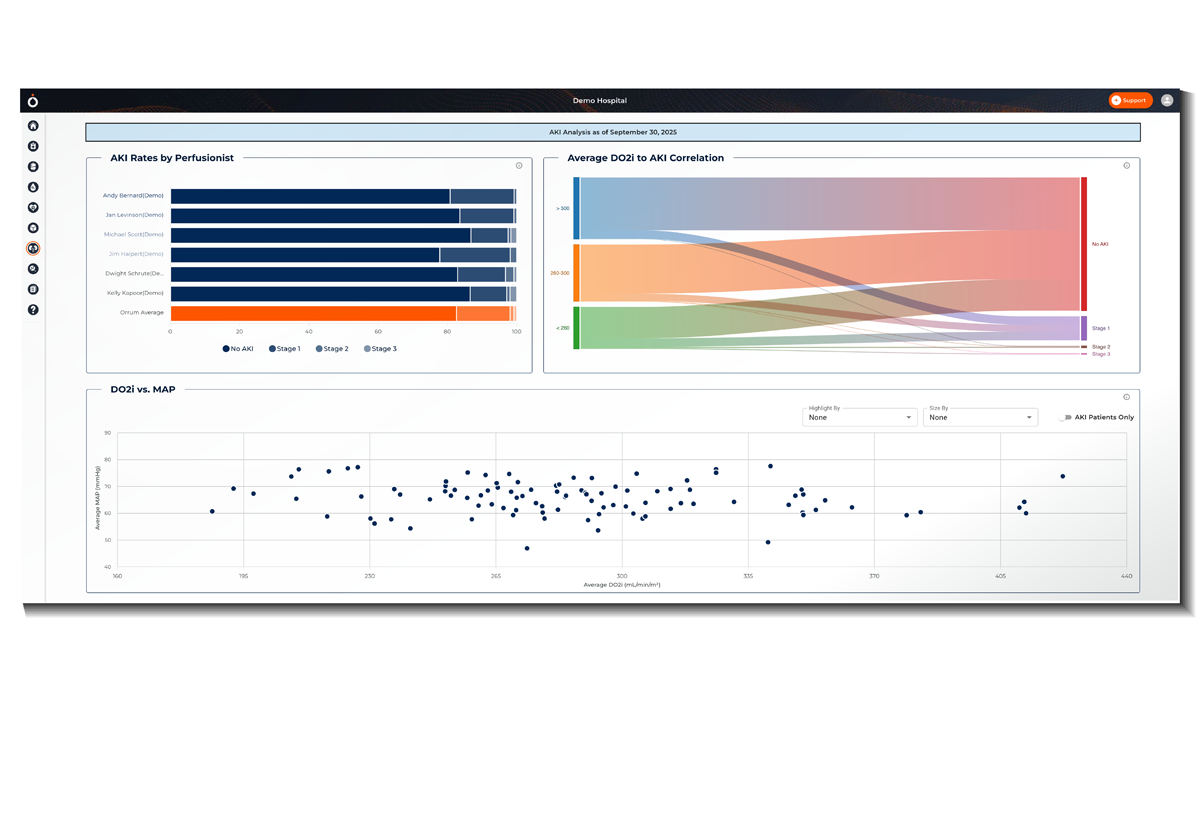
Benchmark performance against peer organizations
Analyze standardized benchmarking data to assess your team’s performance on various perfusion measures compared to other organizations.
Observe local adherence and course correction to evidence-based best practices.
.jpg?language=en-US)
Safety Information
Heart-Lung Machine with Integrated Essenz In-Line Blood Monitor (ILBM)
The Essenz Heart-Lung Machine is intended to perform, control, monitor and support extracorporeal blood circulation replacing the mechanical pumping function of the heart, monitoring and regulating physiologic parameters during procedures requiring extracorporeal circulation.
US: Essenz HLM is intended to be used during cardiopulmonary bypass for procedures lasting six (6) hours or less.
Essenz ILBM is indicated for supplementary, in-line monitoring of the extracorporeal arterial oxygen partial pressure, venous oxygen saturation, venous hematocrit/hemoglobin, and arterial and venous temperature during cardiopulmonary bypass procedures up to six hours.
Essenz Patient Monitor
The Essenz Patient Monitor software is a modularly structured software program package that is exclusively used with LivaNova heart-lung machines. The system allows detailed recording of perfusion data during cardiopulmonary bypass procedures as well as the processing and evaluation of this data. The data may be recorded automatically or entered manually. The LivaNova Perfusion System Monitor is a panel PC intended to be exclusively used with LivaNova heart lung machines as a base and user interface for the Essenz Patient Monitor software. Essenz Patient Monitor is an FDA Cleared medical device.
CONTRAINDICATIONS: No contraindications are known if the devices are used for the purpose described and in accordance with the stated operating conditions. Do not use the devices for any purpose other than indicated.
WARNINGS: The devices must be used in accordance with the instructions for use provided. For a complete listing of warnings please refer to the Instructions for Use. Medical intervention and therapeutic procedure must not be based solely on the Essenz Patient Monitor perfusion records (reports).
PRECAUTIONS: Federal law (U.S.A.) restricts these devices to sale by or on the order of a physician. For a complete listing of precautions/cautions please refer to the Instructions for Use. The devices should be used by qualified and skilled personnel, able to follow the indications and instructions for use contained in the information provided by the manufacturer.
Essenz In-Line Blood Monitor and B-Capta Blood Gas Monitor are clinically equivalent in terms of safety and performance as they have the same technology and intended use. Not approved in all geographies, consult your labeling. Please visit the LivaNova website to receive instructions for use containing full prescribing information including indications, contraindications, warnings, precautions and adverse.
Legal Manufacturers:
LivaNova Deutschland
Lindberghstrasse 25
D-80939
Munich, Germany
Sorin group italia S.r.l.
Via Statale 12 Nord, 86
Mirandola (MO) Italy
Distributed in the US by:
LivaNova USA
14401 W 65th Way
Arvada, CO 80004, USA
CoreInsights
CoreInsights is a service curated by Orrum PSO, a Patient Safety Organization (PSO), with parent organization Orrum Clinical Analytics.
CoreInsights developed by
Orrum Clinical Analytics
45207 Helm St
Plymouth, MI 48170