The ProtekDuo+™ Cannula is Now Available
New product enhancements and MDR certified
Experience the next generation of the ProtekDuo™ cannula
Order codes:
ProtekDuo+ 29 Fr | Order Code: 5150-4629EU
ProtekDuo+ 31 Fr | Order code: 5150-5131EU
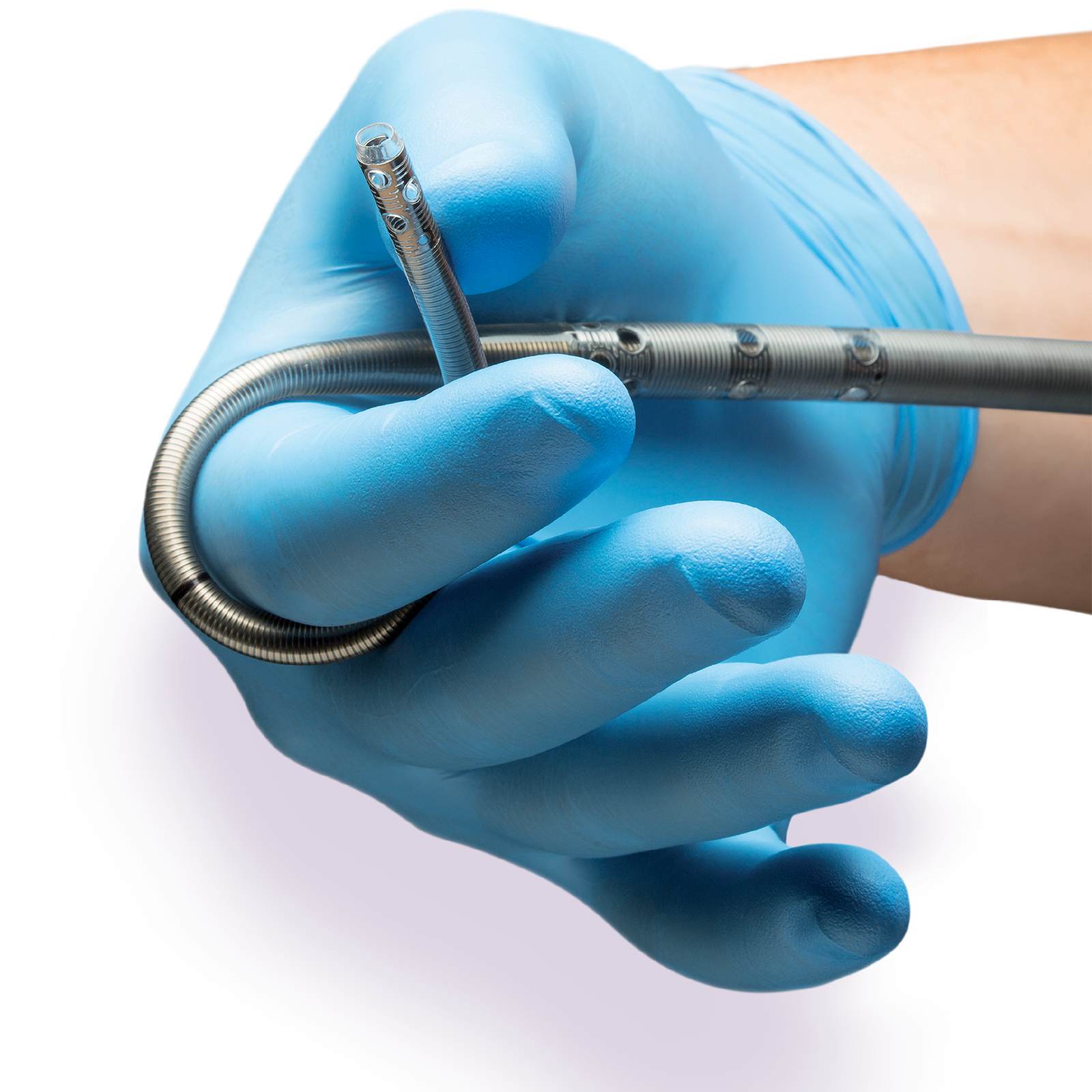
New Product Enhancements
We’ve added user-friendly design and biocompatibility enhancements to the proven cannula with a 10-year clinical legacy.

Color Hemostasis Caps

White Printing on Clamping Zone
Intended to make it easier to read while there is blood in the cannula.

Fewer Depth Insertion Markers
Intended to reduce the patient’s internal exposure to the ink on the cannula body.

Dual by design, multiple by applications
The dual lumen RA-PA cannula design of ProtekDuo+:
- Minimizes recirculation due to the distance between inflow and outflow ports
- Unloads the RV offering partial hemodynamic support
- Increases flexibility in adapting the therapy strategy depending on patient conditions
- Requires only one percutaneous groin-free venous access, improving patient quality of life while on support (patient mobilization and awake management)
How ProtekDuo+ Works
Introducing ProtekDuo+ Dual Lumen Cannula
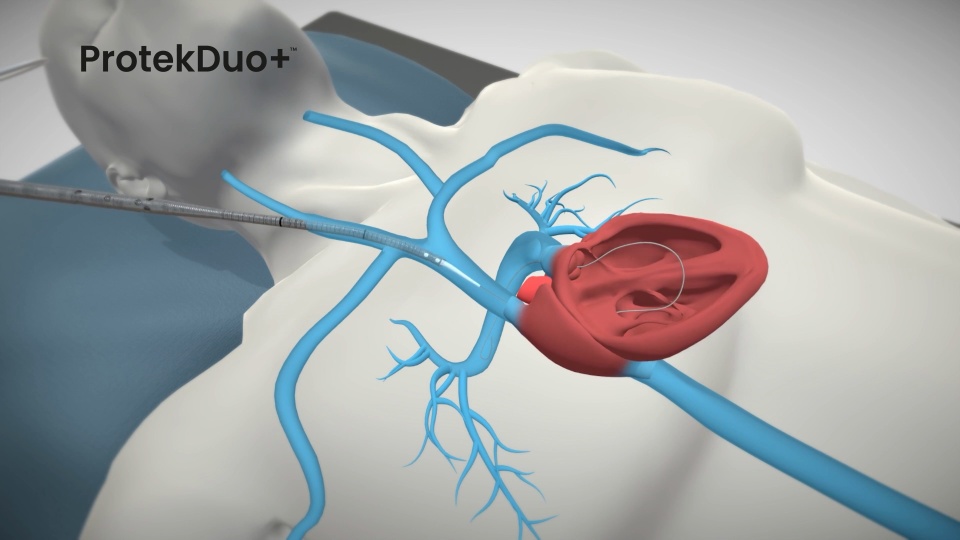
How to Insert ProtekDuo+
Resources
Technical & Ordering Info of the ProtekDuo+
| Sizes | 29 Fr Cannula: Proximal Lumen 29 Fr x 28 cm Distal Lumen 16 Fr x 46 cm Introducer 13 Fr Two suture wings 31 Fr Cannula: Proximal Lumen 31 Fr x 28 cm Distal Lumen 18.5 Fr x 51 cm Introducer 15.5 Fr Two suture wings |
| Usage | Single Use Only |
| Sterilization | Ethylene Oxide Gas |
| Latex | It does not contain latex |
| Insertion accessories needed (not supplied) |
|
| Order numbers | 29 Fr: 5150-4629EU 31 Fr: 5150-5131EU |
| Catalog pages | 29 Fr 31 Fr |
Important Safety Information for ProtekDuo+™ Cannula
Summary of Safety & Performance Information for ProtekDuo+™
INDICATIONS FOR USE/INTENDED USE
Indications for Use
The ProtekDuo+ cannula sets are intended only for use in patients requiring ECLS support with large enough venous access to accommodate the 29 Fr or 31 Fr cannula. Patient condition will vary from healthy (but with high risk of a pulmonary and/or cardiac event) to critically ill, potentially near death. The cannula serves as an essential component of an extracorporeal life support system. The system is deployed to support critically ill patients where other treatment options have failed and continued clinical deterioration is expected or the risk of death is imminent.
Intended Purpose
ProtekDuo+ Cannula sets are single use devices intended for percutaneous drainage and reinfusion of blood to supplement right ventricular cardiac output with or without the use of supplemental extracorporeal membrane gas exchange
CONTRAINDICATIONS
The cannula and introducer are not intended as stand-alone medical devices. There are no known contraindications for the use of the cannula, other than those generally contraindicated for cardiopulmonary bypass. The cannula introducer is only to be used with the appropriately sized ProtekDuo+ cannula. These devices are not intended for use except as indicated above.
WARNINGS
The device must be used in accordance with the instructions for use provided. For a complete listing of warnings please refer to the Instructions for Use which accompany each product
PRECAUTIONS
Federal law (U.S.A.) restricts this device to sale by or on the order of a physician. For a complete listing of precautions/cautions please refer to the Instructions for Use which accompany each product
ADVERSE EVENTS
Potential adverse effects that may be associated with venous cannulation include the following:
Injury to or perforation of the myocardial wall with or without cardiac tamponade; Thrombus formation; Particulate or air embolism; Myocardial infarction; Pulmonary embolism; Cardiac arrhythmias such as atrial fibrillation, heart block, sinus bradycardia and ventricular tachycardia or fibrillation; Congestive heart failure or pulmonary edema; Atrial/ventricular septal defect, transient or persistent, with or without hemodynamic compromise; Vascular injury with or without the need for surgical intervention; Blood loss requiring fluid replacement or transfusion of blood products; Infection; Allergy or anaphylactic reaction to contrast media or device components; Respiratory arrest; Renal failure; Death; Failure to traverse the vascular system; Hemolysis; Pneumothorax; Neurological dysfunction¸ Ischemic stroke¸ Superior Vena Cava Syndrome: characterized by cyanosis, edema, and neurologic sequelae, resulting from venous occlusion.
PERFORMANCE INFORMATION
The devices are designed to come into contact with patient blood and are single use, non-toxic, non-pyrogenic, supplied sterile in individual packaging. Sterilised by ethylene oxide. The level of ethylene oxide residuals in the device is within the limits established by national regulations in the country of use.
The devices should be used by qualified and skilled personnel, able to follow the indications and instructions for use contained in the information provided by the manufacturer. Please contact us through our website (www.sorinmanuals.com) to receive instructions for use containing full prescribing information including indications, contraindications, warnings, precautions and adverse events.
The information contained in this summary represents partial excerpts taken from the product labeling. The information is not intended to serve as a substitute for a complete and through understanding of the device nor does this information represent full disclosure of all pertinent information concerning the use of this product.
Legal Manufacturer:
Sorin Group Italia S.r.l.
Via Statale 12 Nord, 86
41037 Mirandola (MO) Italy
Tel: +39 0535 29811