LifeSPARC™

What's Included
LifeSPARC System Components
Controller
Streamlined and Portable
The lightweight controller features four simple screens and visual and audio alarms. It can be mounted to an IV pole or removed from the docking station for in-hospital transport.

Pump
Simplified Patient Management
The system features the world’s only sterile on-patient pump, which reduces circuit surface area to decrease inflammatory response and eliminate the need for a heater/cooler.

Priming Tray
Rapid Initiation of Support
The unique sterile priming basin uses gravity for a rapid, repeatable priming process. This plug-and-play design allows the system to be primed within 5 minutes.

LifeSPARC Controller
Technical Features
| DIMENSIONS |
H 30.48 cm (12 in) W 21.59 cm (8.5 in) D 20.32 cm (8 in) |
| WEIGHT | 16.5 lbs. (7.48 kg) |
| INTERFACE | Four loop panel GUI |
| ALARMS | Mulitple visual and audio indications |
| POWER SOURCE |
AC power (docking station) Removable, rechargeable Lithium ion batteries |
LifeSPARC Pump
Technical Features
| Flow |
5 LPM percutaneous 8 LPM surgical |
| Speed | 2,000 - 7,500 RPMs |
| Max Pressure | 600 mmHg at 7,500 RPM |
| Bearing | Magnetic pivot bearing |
| Pump Body Priming Volume | 16 mL |
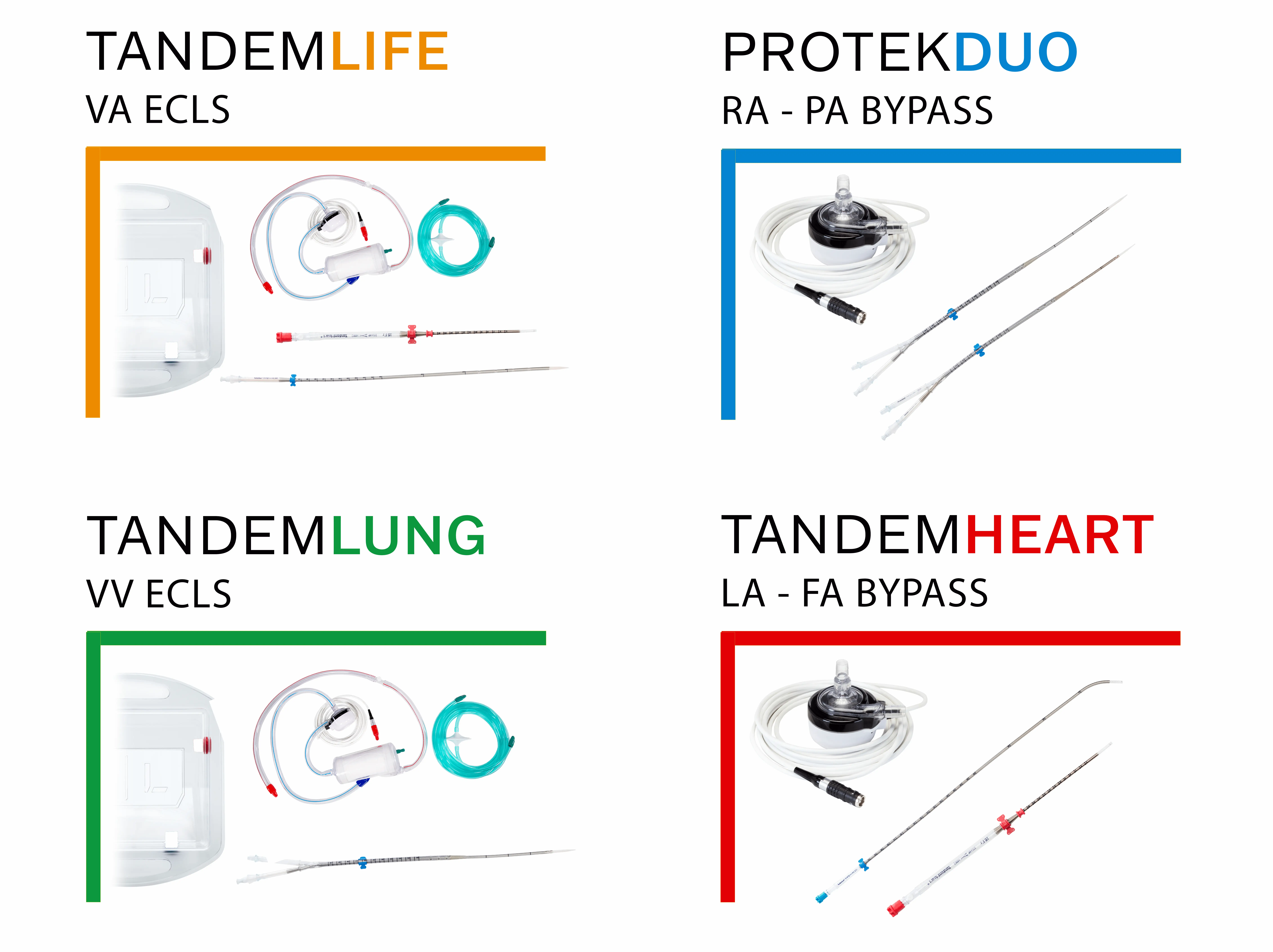
LifeSPARC Procedure Kits
The four ready-to-deploy kits are designed to take the complexity out of emergent rescue. They are turn-key tools that support a variety of cannulation strategies.